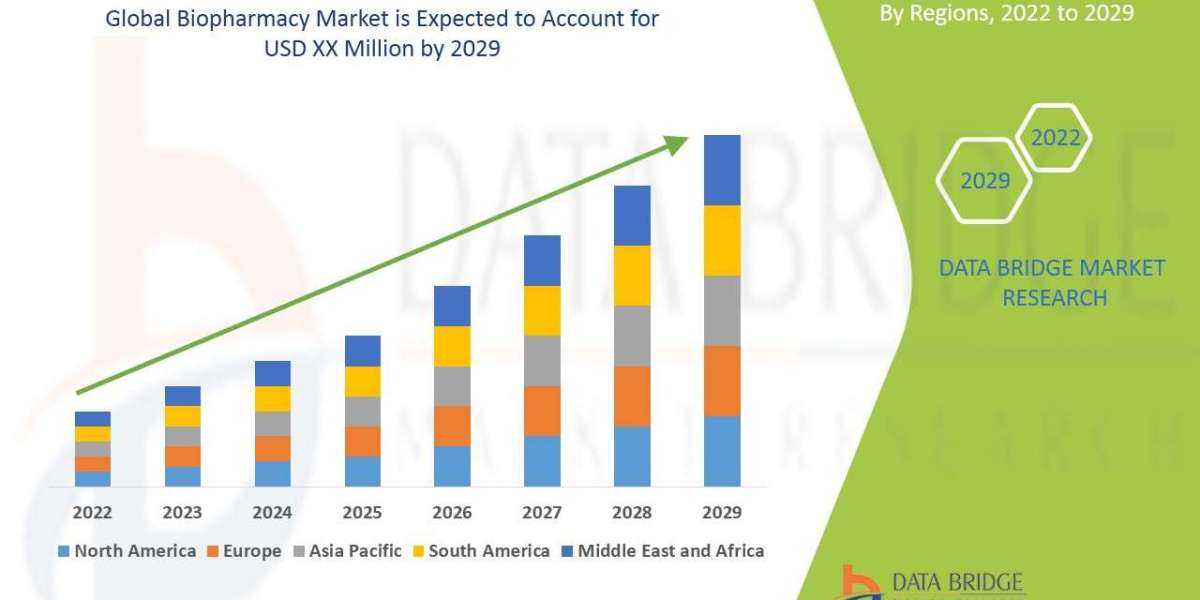
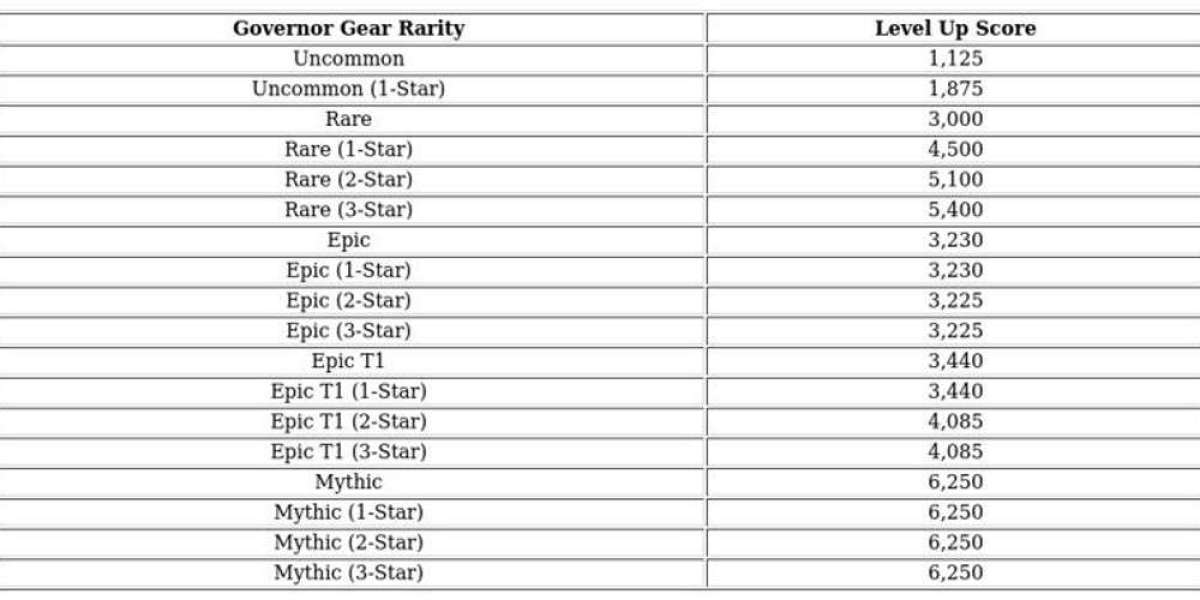
After years of fluctuating perceptions and controversy, Ivermectin doctor support U.S. 2025 has made a surprising comeback. Across the United States, doctors and researchers are revisiting this antiparasitic drug due to accumulating clinical data, growing patient demand, and viral media discussions. The renewed interest is fueled by its anti-inflammatory properties, observed efficacy in reducing COVID-19 symptoms, and its potential role in managing post-COVID syndromes such as long COVID fatigue.
This blog dives into the multifaceted reasons behind Ivermectin’s resurgence — from debates on off-label prescription ethics led by the AMA, to frontline physicians’ testimonials that have spread widely online. We also explore how private clinics in Texas and Florida are pioneering new treatment protocols, and how doctors are expanding their toolkits by revisiting related repurposed drugs like Niclosamide and Fenbendazole, reflecting broader U.S. treatment trends with old antivirals.
⚖️ AMA Debates Off-Label Prescription Ethics: A Turning Point for Medical Practice
In 2025, the American Medical Association (AMA) is engaged in a crucial debate regarding the ethics of off-label prescribing — the practice of using approved medications for non-approved indications. Ivermectin’s comeback has put this issue under a microscope.
- Historically, off-label prescribing is common and often necessary in medicine, allowing flexibility to treat conditions when no approved drugs exist.
- However, given Ivermectin’s controversial history during the pandemic, the AMA is carefully weighing the balance between doctor autonomy and patient safety.
- Some AMA members argue that, with emerging evidence and significant patient interest, doctors should have the liberty to prescribe Ivermectin responsibly based on the latest science.
- Conversely, others stress the importance of waiting for large-scale randomized controlled trials before broad endorsement to avoid premature use or misinformation.
- These ethical discussions are shaping prescribing norms, influencing not only Ivermectin use but broader drug repurposing efforts in the post-pandemic era.
Ultimately, the AMA’s position will affect physician prescribing behavior and public trust in medical guidance for years to come.
? Physicians Cite Anti-Inflammatory Properties in Studies: Scientific Backing Grows
One of the strongest reasons physicians are revisiting Ivermectin is its anti-inflammatory potential, which is critical because uncontrolled inflammation drives much of COVID-19’s severity and lingering symptoms.
- Clinical studies conducted in 2024–2025 show Ivermectin’s ability to reduce levels of cytokines such as IL-6, TNF-alpha, and other inflammatory markers implicated in cytokine storm syndromes.
- This modulation can ease pulmonary inflammation, potentially preventing progression to severe respiratory failure in acute infection and reducing chronic symptoms in long COVID.
- Many doctors report seeing improvements in patients with persistent cough, fatigue, and brain fog after incorporating Ivermectin into treatment regimens.
- Notably, these anti-inflammatory effects complement Ivermectin’s antiviral mechanisms, offering a dual benefit rarely seen in repurposed antivirals.
This growing body of evidence is shifting physician opinion towards supporting Ivermectin as part of a multi-modal treatment plan, especially for patients with persistent symptoms. This shift is reflected in increasing U.S. use and greater calls for responsible clinical support of the drug.
? Rise of Integrative Medicine and Repurposed Drugs: Changing the Treatment Landscape
The healthcare paradigm is shifting. Integrative medicine, which combines evidence-based conventional care with complementary approaches, is growing rapidly. Within this context, drug repurposing has emerged as a pragmatic strategy to address urgent medical needs without the lengthy process of developing new drugs.
- Ivermectin, Niclosamide, and Fenbendazole are prime examples of this approach — drugs with decades of established safety profiles now being tested for new antiviral and immune-modulating indications.
- Physicians practicing integrative medicine focus on holistic patient care, often combining pharmacological treatments with nutrition, physical therapy, and lifestyle interventions.
- This broadened perspective allows for more personalized, adaptive treatment plans tailored to the unique challenges of long COVID and related conditions.
- The success of these repurposed drugs in clinical settings is encouraging physicians to rethink conventional boundaries between standard and complementary medicine.
Consequently, integrative clinics are becoming important centers for post-COVID treatment innovation. This renewed focus adds weight to the potential of Ivermectin for COVID-19 as a multi-functional therapeutic agent.
? Video Testimonials by Frontline Doctors Go Viral: Amplifying Physician Voices
In 2025, social media and video-sharing platforms have transformed medical communication. Frontline doctors, previously confined to academic journals and conferences, now share their real-world clinical experiences directly with the public.
- Video testimonials showcasing doctors describing patient improvements after Ivermectin treatment have garnered millions of views, sparking widespread interest.
- These testimonials often detail patient stories with dramatic symptom relief, ranging from reduced inflammation to faster recovery from fatigue and respiratory issues.
- The transparency and authenticity of firsthand accounts resonate deeply with both patients and other healthcare providers, accelerating dialogue beyond traditional medical circles.
- This viral spread also fuels patient-driven demand for access to Ivermectin, pressuring healthcare systems and regulators to reconsider drug availability.
This digital wave reintroduces Ivermectin into mainstream conversation beyond traditional medical journals, supported by clear information on correct Ivermectin dosage for humans.
? Clinics in Texas and Florida Restart Treatments: A Regional Trend with National Implications
Texas and Florida have emerged as pioneering states for the renewed use of Ivermectin in COVID-19 care. Several private clinics have restarted prescribing the drug, responding to both new data and patient interest.
- These clinics emphasize strict adherence to scientifically supported Ivermectin dosage for humans, usually ranging from Ivermectin 6mg to Ivermectin 12mg depending on clinical evaluation.
- Physicians in these states report increased patient satisfaction and measurable clinical improvements, particularly in managing long COVID symptoms such as fatigue and respiratory distress.
- The local political and regulatory climate also tends to support more flexible prescribing practices, allowing clinics to explore innovative treatments faster than in other states.
- Clinics have incorporated Ivermectin into multi-drug protocols alongside vitamins and supportive therapies, observing synergistic effects.
The experiences of Texas and Florida clinics may influence prescribing patterns nationwide, potentially guiding federal policy adaptations.
? Patient Survey Data Shows Satisfaction Trends: Real-World Evidence Emerges
Recent surveys conducted by clinics prescribing Ivermectin provide valuable real-world insights into patient outcomes:
- More than 75% of surveyed patients reported meaningful improvements in symptoms like chronic fatigue, muscle aches, and brain fog within weeks of starting treatment.
- Patient feedback highlights the psychological benefit of having active treatment options in the face of limited official recommendations.
- Reports of adverse effects remain low and mostly mild, with few discontinuations due to side effects.
- Survey data is helping clinics refine dosing protocols and identify patient subgroups who benefit most.
These findings help bridge the gap between clinical trials and everyday practice, fostering confidence in Ivermectin’s role in post-COVID care.
? Doctors Also Revisit Niclosamide and Fenbendazole Efficacy: Expanding the Therapeutic Arsenal
While Ivermectin headlines much of the discussion, physicians are also revisiting two other repurposed drugs — Niclosamide and Fenbendazole — known for their antiviral and immune system effects.
- Niclosamide inhibits viral replication and modulates immune responses, showing promise in reducing symptom duration and severity.
- Fenbendazole, used primarily in veterinary medicine, has demonstrated immunomodulatory and autophagy-enhancing properties, potentially aiding in the clearance of viral remnants and damaged cells.
- Combined with Ivermectin, these drugs are being evaluated in multi-agent treatment protocols to tackle COVID-19’s complex pathophysiology from multiple angles.
- Early clinical experiences suggest this approach may improve outcomes in stubborn long COVID cases resistant to standard care.
Continued research is crucial to define optimal regimens and long-term safety profiles for these combination therapies.
❓ Frequently Asked Questions (FAQ)
Q1: Why are doctors revisiting Ivermectin in 2025?
A1: Because new research and clinical observations highlight its anti-inflammatory and antiviral benefits, alongside strong patient demand and frontline physician support.
Q2: What ethical issues surround off-label prescribing of Ivermectin?
A2: The AMA debates balancing physician freedom to innovate with the need for robust scientific validation and patient safety.
Q3: What is the typical Ivermectin dosage for treating COVID-19?
A3: Dosages usually range from 6mg to 12mg, tailored by doctors based on individual patient profiles.
Q4: Is buying Ivermectin online safe?
A4: It can be, if sourced from reliable providers like Medicoease, which ensures product quality and appropriate dosing.
Q5: Are Niclosamide and Fenbendazole viable alternatives or supplements?
A5: Preliminary data are encouraging, suggesting they can be effective when used alongside Ivermectin in multi-drug protocols.
Q6: How quickly can patients expect improvement?
A6: Some report benefits within 2-4 weeks, though response times vary with severity and individual health factors.
? Conclusion: A New Chapter for Ivermectin and Repurposed Drugs in U.S. Medicine
The spotlight on Ivermectin for COVID-19 in 2025 reflects the dynamic nature of medical practice adapting to emerging science and patient needs. Physicians nationwide are reevaluating old drugs in light of new data and integrative care trends, embracing a personalized, multi-faceted approach to COVID-19 treatment and recovery.
Clinics in Texas, Florida, and beyond are pioneering safe, effective protocols, supported by real-world patient satisfaction and physician advocacy. The renewed interest in Niclosamide and Fenbendazole further expands therapeutic options against complex viral and inflammatory conditions.
For patients seeking treatment, Medicoease remains the trusted platform to buy ivermectin online USA with guaranteed quality and medical oversight.
As the medical community continues to balance innovation with evidence-based care, Ivermectin and its repurposed counterparts may hold an important place in the evolving landscape of COVID-19 therapy.
For a comprehensive background on Ivermectin and related drugs, visit Wikipedia.